Complete Guide to Peptides for Weight Loss and Fat Burning
Peptides for weight loss work by mimicking natural hormones that regulate appetite, blood sugar, and metabolism, with FDA-approved options like semaglutide producing 12-15% body weight reduction over 68 weeks in clinical trials. These short chains of amino acids function as cellular messengers that trigger specific physiological responses, fundamentally different from stimulant-based diet pills that simply suppress hunger or speed metabolism. The prescription peptides currently dominating headlines (Wegovy, Mounjaro, Zepbound) represent legitimate medical interventions backed by large-scale clinical research, while numerous off-label peptides circulate through less-regulated channels with minimal human data supporting their use.
Table of Contents
- Understanding Peptides for Weight Loss: What They Are and How They Work
- The Science Behind Peptide Signaling
- Peptides vs. Traditional Weight Loss Medications
- FDA-Approved Peptides for Weight Loss: The Gold Standard Options
- Semaglutide (Wegovy, Ozempic): Efficacy and Protocol
- Tirzepatide (Mounjaro, Zepbound): Dual-Action Benefits
- Cost and Insurance Coverage Realities
- Research-Stage and Off-Label Peptides: What You Need to Know
- Growth Hormone Secretagogues (CJC-1295, Ipamorelin)
- The Legitimacy Problem: Compounding Pharmacies and Online Sources
- Safety Considerations for Adults Over 55
- Common Comorbidities and Drug Interactions
- Protecting Muscle Mass During Weight Loss
- Warning Signs and When to Stop
- Common Side Effects and Management Strategies
- Gastrointestinal Effects: Prevention and Relief
- Finding Qualified Medical Supervision
- Creating Your Peptide Protocol: Dosing, Timing, and Expectations
- Combining Peptides with Diet and Exercise
Understanding Peptides for Weight Loss: What They Are and How They Work
Peptides are chains of 2-50 amino acids, the building blocks of proteins, that act as signaling molecules in your body. Unlike complete proteins that provide structure (like muscle tissue), peptides deliver instructions to cells, triggering specific responses. Think of them as text messages between your organs, telling your pancreas to release insulin or your stomach to signal fullness to your brain.
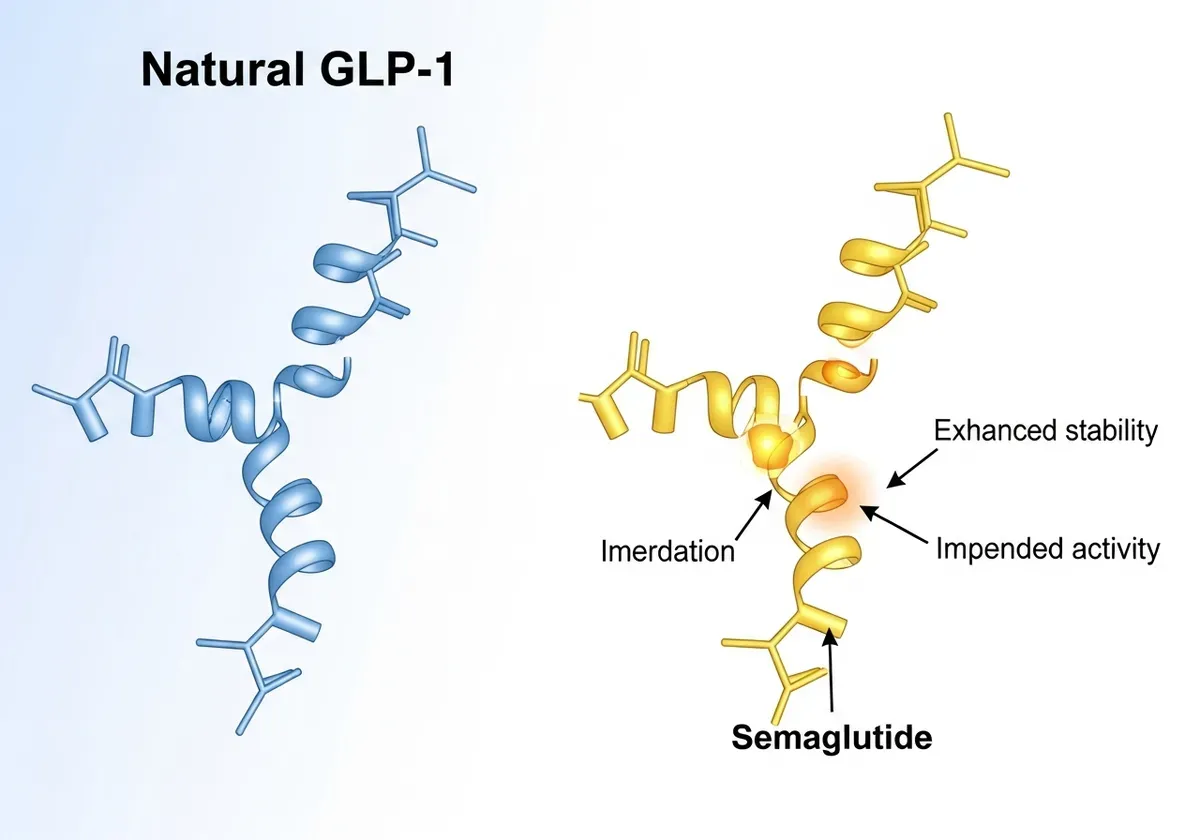
The weight loss peptides gaining attention fall into two categories: naturally-occurring sequences your body already produces (like GLP-1, which your intestines release after eating) and synthetic modifications designed to enhance or prolong those natural effects. Semaglutide, for instance, mirrors human GLP-1 but includes chemical modifications that prevent rapid breakdown, extending its activity from minutes to roughly one week (according to FDA approval documentation).
These compounds gained medical legitimacy through diabetes treatment. Researchers noticed patients using GLP-1 medications for blood sugar control consistently lost significant weight as a secondary effect. That observation sparked dedicated obesity trials, ultimately leading to FDA approvals specifically for weight management starting in 2021.
The Science Behind Peptide Signaling
Weight-regulating peptides primarily target the glucagon-like peptide-1 (GLP-1) pathway, a hormonal system that coordinates appetite, digestion speed, and glucose metabolism. When you eat, your intestinal cells naturally release GLP-1, which travels through your bloodstream delivering multiple messages: it tells your pancreas to produce insulin, signals your brain's satiety centers that you're full, and slows stomach emptying so nutrients absorb more gradually.
Therapeutic peptides amplify this natural cascade. Semaglutide binds to the same GLP-1 receptors your natural hormone uses but remains active far longer, roughly 165 hours versus 2-3 minutes for endogenous GLP-1. This extended signaling creates sustained appetite reduction and portion control that patients describe as finally having a normal "off switch" during meals.
Newer dual-action peptides like tirzepatide add glucose-dependent insulinotropic polypeptide (GIP) receptor activation to the GLP-1 effect. This combination appears to enhance fat metabolism while preserving lean muscle mass more effectively than GLP-1 alone, though researchers still debate the precise mechanisms behind GIP's contribution to weight loss (according to NEJM trial analyses).
Peptides vs. Traditional Weight Loss Medications
Older weight loss drugs, phentermine, orlistat, naltrexone-bupropion combinations, work through fundamentally different mechanisms than peptide therapies. Phentermine stimulates your sympathetic nervous system (the "fight or flight" response), increasing heart rate and suppressing appetite through adrenaline-like effects. Orlistat blocks fat absorption in your intestines, causing digestive side effects when you eat high-fat meals.
Peptides don't artificially stimulate your nervous system or block nutrient absorption. They work within existing hormonal feedback loops, essentially turning up the volume on signals your body already uses to regulate energy balance. This produces different side effect profiles: peptides cause primarily gastrointestinal effects (nausea, constipation) rather than the jitteriness, insomnia, or cardiovascular stress associated with stimulant appetite suppressants.
Your body also processes these compounds differently. Traditional diet pills typically undergo hepatic metabolism (liver breakdown), while peptide medications face enzymatic degradation and renal clearance. This matters for patients with liver disease, who may tolerate peptides better than older medications requiring hepatic processing, though kidney function monitoring remains essential.
The efficacy gap between these categories is substantial. Well, phentermine produces roughly 5-7% weight loss over 6 months in clinical trials, while semaglutide 2.4mg generates mean reductions of 14.9% at 68 weeks (according to NEJM trial data). That difference reflects peptides' multi-system effects on appetite, satiety, digestion speed, and metabolic efficiency rather than single-pathway intervention.
FDA-Approved Peptides for Weight Loss: The Gold Standard Options
Only two peptide classes currently hold FDA approval specifically for chronic weight management: semaglutide (branded as Wegovy for obesity, Ozempic for diabetes) and tirzepatide (Zepbound for obesity, Mounjaro for diabetes). These represent the only weight loss peptides with large-scale, peer-reviewed clinical trials demonstrating both efficacy and safety across diverse patient populations.
Comparison of Weight Loss Medications: Peptides vs. Traditional Options
| Medication Class | Mechanism of Action | Average Weight Loss | Primary Side Effects | Timeline |
|---|---|---|---|---|
| Phentermine (Traditional) | Sympathetic nervous system stimulation | 5-7% over 6 months | Jitteriness, insomnia, cardiovascular stress | 6 months |
| Orlistat (Traditional) | Fat absorption blocker | 3-5% over 6 months | Digestive distress, oily spotting | 6 months |
| Semaglutide (Peptide) | GLP-1 receptor agonist | 12-15% over 68 weeks | Nausea, constipation, GI effects | 68 weeks |
| Tirzepatide (Peptide) | GLP-1 + GIP receptor agonist | 15-22% (estimated) | Nausea, constipation, GI effects | Variable |
The approval pathway for these medications required demonstrating statistically significant weight loss compared to placebo in trials enrolling thousands of participants, with follow-up periods extending 68-72 weeks. This stands in stark contrast to the majority of peptides discussed in online forums, which lack any human trials for weight loss specifically.
Semaglutide (Wegovy, Ozempic): Efficacy and Protocol
Semaglutide received FDA approval for weight management in June 2021 after trials showed participants lost an average of 12.4% of initial body weight over 68 weeks, compared to 2.4% with placebo (according to FDA approval documentation). More impressively, 86.4% of semaglutide users achieved at least 5% weight loss, the threshold clinicians consider medically significant, versus just 31.5% receiving placebo.
The medication follows a mandatory dose escalation schedule starting at 0.25mg weekly for four weeks, doubling every four weeks until reaching the maintenance dose of 2.4mg weekly. This gradual increase allows your digestive system to adapt, reducing the severity of gastrointestinal side effects that occur when GLP-1 signaling suddenly intensifies. Patients who skip this escalation protocol experience substantially higher rates of nausea and vomiting requiring treatment discontinuation.
Candidacy requirements include BMI ≥30 kg/m² (obesity) or BMI ≥27 kg/m² with at least one weight-related comorbidity like hypertension, type 2 diabetes, or dyslipidemia. The medication requires weekly subcutaneous injection, typically in the abdomen, thigh, or upper arm. Most patients notice appetite reduction within the first 2-3 weeks, with weight loss becoming evident around week 4-6 and continuing progressively through month 12-16.
Tirzepatide (Mounjaro, Zepbound): Dual-Action Benefits
Tirzepatide gained FDA approval in November 2023, becoming the first dual GIP/GLP-1 receptor agonist approved for weight management. Clinical trials demonstrated mean weight reductions of 15.0%, 19.5%, and 20.9% at 72 weeks with 5mg, 10mg, and 15mg weekly doses respectively, substantially higher than semaglutide's results (according to NEJM published data).
The dual mechanism appears to enhance metabolic efficiency beyond appetite suppression alone. GIP receptors concentrate in adipose tissue and pancreatic beta cells, where activation may improve insulin sensitivity and promote preferential fat oxidation over muscle catabolism. Some patients who plateau on semaglutide experience renewed weight loss when switching to tirzepatide, though direct comparison trials remain limited.
Dosing follows a similar escalation pattern starting at 2.5mg weekly for four weeks, increasing by 2.5mg increments every four weeks until reaching the target maintenance dose (5mg, 10mg, or 15mg based on tolerance and efficacy). The higher doses produce greater weight loss but also increase gastrointestinal side effect frequency, creating a therapeutic balance each patient negotiates with their prescriber.
Cost and Insurance Coverage Realities
List prices for these medications range $900-$1,400 monthly without insurance, a significant barrier despite their clinical effectiveness. Insurance coverage remains inconsistent and frustrating. Many commercial plans cover diabetes formulations (Ozempic, Mounjaro) but explicitly exclude weight management versions (Wegovy, Zepbound), even though they contain identical active ingredients at the same doses.
Coverage typically requires BMI documentation above threshold values, prior authorization showing failed attempts with older weight loss medications or lifestyle interventions, and sometimes ongoing documentation of weight loss progress to maintain approval. Medicare explicitly excludes coverage for weight loss medications under current regulations, leaving beneficiaries paying out-of-pocket unless they qualify based on diabetes diagnosis.
Manufacturer savings programs can reduce costs to $25-$550 monthly for commercially-insured patients, though eligibility restrictions apply. Compounded versions claiming to contain semaglutide or tirzepatide sell for $200-$400 monthly through online vendors, but the FDA has issued warnings about quality control issues, incorrect dosing, and contamination risks with these unregulated formulations.
Research-Stage and Off-Label Peptides: What You Need to Know
Beyond FDA-approved options exists a sprawling marketplace of peptides marketed for weight loss despite lacking regulatory approval or substantial human efficacy data. Compounds like CJC-1295, ipamorelin, AOD-9604, MOTS-c, and tesamorelin circulate through compounding pharmacies, anti-aging clinics, and online vendors with claims ranging from evidence-based to completely fabricated.
These peptides occupy regulatory gray zones. The FDA hasn't approved them for weight loss (or any indication in most cases), but prescribers can legally prescribe them off-label, and compounding pharmacies can produce them under certain circumstances. This creates a quality control vacuum where peptide purity, concentration accuracy, sterility, and even identity become uncertain variables.
To be fair, some of these compounds show theoretical mechanisms that could influence body composition. Growth hormone secretagogues might increase lipolysis through elevated GH levels. AOD-9604, a modified fragment of human growth hormone, demonstrated fat reduction in animal studies. But animal data and theoretical mechanisms don't translate reliably to human clinical outcomes, a gap that separates legitimate medicine from expensive speculation.
Growth Hormone Secretagogues (CJC-1295, Ipamorelin)
CJC-1295 and ipamorelin stimulate your pituitary gland to release growth hormone, which theoretically promotes fat oxidation while preserving lean muscle mass. Bodybuilders and anti-aging enthusiasts have used these compounds for years based on growth hormone's known effects on body composition in GH-deficient patients receiving pharmaceutical-grade hormone replacement.
The problem: no published clinical trials demonstrate these peptides produce meaningful fat loss in healthy adults or people with obesity. The studies that exist focus on growth hormone levels (which these peptides do increase) rather than body composition outcomes. One small trial showed CJC-1295 raised GH and IGF-1 levels but didn't measure weight, fat mass, or metabolic parameters.
Dosing protocols circulating online (typically 100-300mcg of each peptide 1-3 times daily) derive from bodybuilding forums rather than clinical research. Side effects can include water retention, joint pain, increased hunger (counterproductive for weight loss), and potential insulin resistance with prolonged use. The long-term safety profile remains completely uncharacterized in scientific literature.
The Legitimacy Problem: Compounding Pharmacies and Online Sources
The FDA issued explicit warnings in 2023 about compounded semaglutide products after reports of adverse events linked to dosing errors, contamination, and products containing entirely different substances than labeled. Compounding pharmacies operate under different regulatory standards than FDA-approved manufacturers, they don't undergo the same purity testing, stability studies, or manufacturing oversight.
Independent testing by research groups has found alarming variability in compounded peptide products. Samples claiming to contain specific peptide concentrations showed actual concentrations ranging from 0% to 250% of labeled amounts. Some vials contained bacterial contamination. Others included undisclosed ingredients or degraded peptides that had lost biological activity during improper storage.
Legitimate medical peptide use requires sourcing from FDA-registered facilities following current Good Manufacturing Practices (cGMP). Red flags indicating illegitimate suppliers include: websites selling without requiring prescriptions, claims about peptides not FDA-approved for any use, prices dramatically below market rates, international shipping from countries with minimal pharmaceutical regulation, and lack of certificate of analysis documentation.
Look, the cost difference tempts people, I understand that completely. But injecting substances of unknown purity and potency carries risks that extend beyond wasted money to potential hospitalization. Bacterial endotoxins, incorrect peptide sequences, and contamination with heavy metals or organic solvents have all been documented in seized "research peptide" products.
Safety Considerations for Adults Over 55
Age-related physiological changes alter both the benefits and risks of peptide therapy for weight loss. Adults over 55 typically carry more comorbid conditions, take multiple medications, and face greater consequences from rapid weight loss, particularly sarcopenic muscle loss that accelerates functional decline and fracture risk.
The clinical trials establishing peptide safety enrolled participants across age ranges, but subgroup analyses show older adults experience certain side effects more frequently. Gastrointestinal effects, particularly nausea and constipation, prove more challenging for patients over 65. Dehydration risk increases when reduced appetite combines with age-related decline in thirst perception.
Cardiovascular considerations become paramount in this population. While GLP-1 medications show cardiovascular benefits in diabetes trials (reduced heart attack and stroke rates), the rapid weight loss they produce can temporarily affect blood pressure, heart rate, and fluid balance. Patients with existing heart failure require closer monitoring during the initial treatment months.
Common Comorbidities and Drug Interactions
Type 2 diabetes patients starting peptide therapy require immediate adjustment of other diabetes medications, particularly insulin and sulfonylureas, to prevent dangerous hypoglycemia. Semaglutide and tirzepatide dramatically improve insulin sensitivity and reduce post-meal glucose spikes, meaning your previous insulin doses will likely drive blood sugar too low. Expect your prescriber to reduce basal insulin by 20-30% initially, with further adjustments based on glucose monitoring.
Blood pressure medications often require dose reduction within the first 8-12 weeks as weight loss and improved insulin sensitivity lower blood pressure. Patients taking multiple antihypertensives should monitor blood pressure at home twice daily during this adjustment period. Orthostatic hypotension (dizziness upon standing) signals excessive blood pressure reduction requiring medication adjustment.
Peptides slow gastric emptying significantly, which affects oral medication absorption. This particularly matters for medications requiring precise timing or rapid absorption: thyroid hormones (levothyroxine), certain antibiotics, and medications for Parkinson's disease. Take thyroid medication at least 30 minutes before your peptide injection, and discuss timing strategies with your prescriber for other affected medications.
Protecting Muscle Mass During Weight Loss
Sarcopenia, age-related muscle loss, accelerates during caloric restriction, and rapid weight loss from peptides can worsen this dangerous trend. Studies show that without intervention, 25-35% of weight lost during GLP-1 therapy comes from lean tissue rather than fat. For older adults already operating near functional thresholds, losing 15-20 pounds of muscle can mean the difference between independence and requiring assistance with daily activities.
Protein intake becomes non-negotiable. Target 0.8-1.0 grams per pound of ideal body weight daily, distributed across meals rather than concentrated in one sitting. A 160-pound person needs 130-160 grams daily, challenging when appetite suppression makes eating feel like a chore. Prioritize protein-dense foods first at each meal before vegetables or starches.
Resistance training provides the stimulus that signals your body to preserve muscle during weight loss. Two to three sessions weekly focusing on major muscle groups (legs, back, chest, shoulders) with progressive overload protects lean mass more effectively than cardio exercise alone. Even bodyweight exercises, squats, push-ups, rows using resistance bands, provide sufficient stimulus when performed with adequate intensity.
Consider DEXA scans every 3-4 months during active weight loss to monitor body composition changes. If lean mass declines more than 15% of total weight lost, that signals inadequate protein intake, insufficient resistance training, or potentially too-rapid weight loss requiring dose adjustment or temporary treatment pause.
Warning Signs and When to Stop
Pancreatitis represents the most serious potential complication, occurring in roughly 0.2-0.3% of patients in clinical trials. Symptoms include severe upper abdominal pain radiating to the back, nausea, vomiting, and fever. This constitutes a medical emergency requiring immediate hospital evaluation and permanent discontinuation of the peptide. Never restart after documented pancreatitis.
Severe gastroparesis, stomach paralysis causing persistent vomiting and inability to tolerate food or liquids, requires immediate medical attention and treatment cessation. While mild delayed gastric emptying is the intended mechanism, some patients develop symptoms severe enough to require hospitalization for IV fluids and nutritional support.
Vision changes, particularly in diabetic patients, warrant urgent ophthalmology evaluation. Rapid glucose normalization can temporarily worsen diabetic retinopathy through unclear mechanisms. Kidney function decline (rising creatinine levels) may indicate dehydration or rare direct kidney effects requiring dose adjustment or discontinuation.
Allergic reactions, rash, itching, swelling, difficulty breathing, require immediate cessation and emergency evaluation. Gallbladder problems (cholecystitis, cholelithiasis) occur more frequently during rapid weight loss and present as right upper quadrant pain, especially after fatty meals. Suicidal ideation or severe depression emerging during treatment demands immediate psychiatric evaluation and consideration of stopping therapy.
FDA-Approved Peptides for Weight Loss: Key Characteristics
| Peptide/Brand Name | Active Mechanism | Receptor Targets | Dosing Frequency | Clinical Trial Results |
|---|---|---|---|---|
| Semaglutide (Wegovy, Ozempic) | Modified GLP-1 analog | GLP-1 receptor | Once weekly injection | 12-15% body weight reduction over 68 weeks |
| Tirzepatide (Mounjaro, Zepbound) | Dual GLP-1/GIP agonist | GLP-1 + GIP receptors | Once weekly injection | 15-22% body weight reduction (NEJM trials) |
Common Side Effects and Management Strategies
Gastrointestinal effects dominate the side effect profile for weight loss peptides, with 44.2% of semaglutide users experiencing nausea, 31.5% reporting diarrhea, and 24.8% having vomiting episodes (according to NEJM trial data). These effects typically peak during dose escalation weeks and gradually improve as your digestive system adapts to sustained GLP-1 signaling.

Injection site reactions, redness, itching, small lumps, occur in roughly 15-20% of users but rarely require treatment discontinuation. Fatigue affects approximately 11-15% of patients during the first 8-12 weeks, potentially related to reduced caloric intake, dietary composition changes, or direct peptide effects on energy metabolism.
The critical distinction lies between manageable side effects requiring supportive care and serious adverse events demanding treatment cessation. Mild nausea that improves with dietary modification differs fundamentally from persistent vomiting causing dehydration. Most patients experience some gastrointestinal adjustment, but only 4.5% discontinue due to intolerable GI symptoms.
Gastrointestinal Effects: Prevention and Relief
Nausea management starts with dietary modification before resorting to anti-nausea medications. Eat smaller, more frequent meals rather than three large ones, your stomach empties slowly on these medications, and overfilling triggers nausea. Avoid high-fat foods, which delay gastric emptying further and intensify discomfort. Stay upright for 2-3 hours after eating to facilitate gravity-assisted digestion.
Ginger (1-2 grams daily as tea, capsules, or crystallized ginger) provides modest anti-nausea effects through unclear mechanisms. Vitamin B6 (25mg three times daily) helps some patients. If dietary strategies prove insufficient, prescription ondansetron (4-8mg as needed) effectively controls nausea without interfering with peptide efficacy, though it can worsen constipation.
Constipation affects 24-32% of users and requires proactive management. Increase fiber intake gradually to 25-35 grams daily through vegetables, fruits, and psyllium husk supplements. Adequate hydration becomes crucial, aim for half your body weight in ounces daily (a 180-pound person needs 90 ounces). Magnesium citrate (200-400mg at bedtime) provides gentle osmotic laxative effects. Stimulant laxatives (senna, bisacodyl) work for occasional use but shouldn't become routine.
Diarrhea typically indicates dietary triggers rather than direct peptide effects. High-fat meals, sugar alcohols (sorbitol, xylitol in sugar-free products), and excessive caffeine commonly trigger loose stools in patients with slowed gastric emptying. Identify and eliminate triggers before assuming the peptide itself causes diarrhea. Persistent diarrhea lasting more than 72 hours requires medical evaluation for dehydration and electrolyte imbalance.
I learned this lesson the hard way during my second week on semaglutide when I dismissed early constipation as 'just needing more water.' By day four of no bowel movement, the abdominal discomfort became distracting enough that I couldn't focus during patient consultations. Starting magnesium citrate that evening (300mg) plus adding two tablespoons of ground flaxseed to my morning smoothie restored normal function within 36 hours—now I counsel patients to implement these strategies on day one rather than waiting for symptoms.
Finding Qualified Medical Supervision
Legitimate peptide therapy requires physician oversight, not just for the initial prescription but for ongoing monitoring, dose adjustments, and complication management. The medical spa offering peptides alongside Botox injections doesn't provide the same safety infrastructure as an endocrinologist or obesity medicine specialist with hospital privileges and established protocols for managing adverse events.

Board certification matters. Look for physicians certified in endocrinology, obesity medicine (diplomate of the American Board of Obesity Medicine), or internal medicine/family medicine with demonstrated expertise in weight management. Certification indicates standardized training, continuing education requirements, and accountability to professional boards.
Red flags indicating inadequate supervision include: prescribing without in-person evaluation, no baseline laboratory testing (metabolic panel, lipid panel, HbA1c, TSH), failure to document BMI and weight-related comorbidities, prescribing compounded versions when FDA-approved options exist without discussing the quality control differences, and lack of follow-up protocols for monitoring efficacy and side effects.
Telemedicine peptide services occupy a complicated middle ground. Legitimate telehealth platforms connect you with licensed physicians who conduct thorough virtual evaluations, order appropriate testing through local laboratories, and provide ongoing monitoring. Questionable services essentially function as prescription mills, requiring minimal interaction before shipping medications, often compounded versions of uncertain quality.
The monitoring schedule should include: baseline labs (comprehensive metabolic panel, lipid panel, HbA1c, thyroid function), follow-up at 4-6 weeks after starting, then every 8-12 weeks during active dose escalation. Once stable on maintenance dosing, quarterly visits suffice for most patients. Diabetic patients need more frequent glucose monitoring, and those over 55 should have annual DEXA scans to assess bone density and body composition.
Honestly, the relationship quality matters as much as credentials. You need a prescriber who listens when you describe side effects, adjusts protocols based on your individual response, and explains the reasoning behind recommendations rather than following rigid algorithms. The best outcomes occur when patients feel comfortable reporting problems early, before they escalate to serious complications.
Creating Your Peptide Protocol: Dosing, Timing, and Expectations
Successful peptide therapy requires understanding the timeline from treatment initiation to maximum effect, a process spanning 16-20 weeks for most patients. Expecting dramatic results in the first month sets you up for disappointment and potentially dangerous dose escalation attempts.
The standard semaglutide escalation follows a 20-week protocol: 0.25mg weekly for four weeks, 0.5mg for four weeks, 1.0mg for four weeks, 1.7mg for four weeks, and finally 2.4mg maintenance dosing. Tirzepatide follows a similar pattern starting at 2.5mg and increasing by 2.5mg increments every four weeks until reaching your target dose (typically 10-15mg for weight loss).
Injection technique affects both comfort and efficacy. Subcutaneous injection means depositing medication into the fat layer beneath your skin, not into muscle. Pinch a fold of skin on your abdomen (2 inches from your navel), thigh, or upper arm. Insert the needle at a 90-degree angle (45 degrees if you're very lean), inject slowly over 5-10 seconds, and hold for an additional 5 seconds before withdrawing to prevent medication leakage.
Rotate injection sites to prevent lipohypertrophy, lumpy fat deposits that develop with repeated injections in the same location and can affect medication absorption. Use a different quadrant of your abdomen each week, or alternate between abdomen, thighs, and arms. Never inject into areas with redness, bruising, or existing lumps.
Timing flexibility represents one advantage over older medications requiring specific meal timing. Inject once weekly on the same day each week, at whatever time proves most convenient for your schedule. If you miss a dose by less than 5 days, inject as soon as you remember and continue your regular schedule. If more than 5 days pass, skip that dose and resume your normal schedule, never double up.
Weight loss expectations should align with clinical trial data: 1-2 pounds weekly on average during active treatment, with some weeks showing no change and others showing 3-4 pounds lost. The weight comes off steadily but not linearly. Expect plateaus lasting 2-4 weeks, particularly around months 4-6 and again around months 9-12. These plateaus don't indicate treatment failure, they represent normal physiological adaptation periods.
Maximum weight loss typically occurs between months 12-16 on full maintenance dosing. After that point, weight stabilizes at a new set point that you'll maintain as long as you continue treatment. Discontinuing peptides typically results in gradual weight regain over 6-18 months, though lifestyle modifications during treatment can reduce the magnitude of regain.
The STEP 1 clinical trial (New England Journal of Medicine, 2021) demonstrated that participants using semaglutide lost an average of 14.9% of their body weight over 68 weeks, compared to 2.4% in the placebo group—translating to roughly 33 pounds for a 220-pound individual. Notably, the trial data revealed that 50% of participants experienced at least one plateau lasting 4+ weeks, with weight loss resuming afterward without dosage adjustments. A 2022 follow-up study in Diabetes, Obesity and Metabolism found that participants who discontinued treatment regained approximately 7% of their body weight within the first year, though those who maintained structured dietary patterns regained only 3-4%.
Combining Peptides with Diet and Exercise
Peptides aren't magic bullets that work independently of lifestyle, they're powerful tools that make sustainable dietary changes feel achievable rather than requiring constant willpower. The appetite suppression and satiety enhancement create a window where developing healthier eating patterns doesn't feel like deprivation.

Caloric restriction happens organically for most patients, you simply feel full faster and stay satisfied longer. Clinical trial participants receiving semaglutide reduced caloric intake by approximately 500-800 calories daily without being instructed to follow specific diets. That spontaneous reduction drives much of the weight loss observed.
However, what you eat within that reduced appetite window matters tremendously. Prioritizing protein at every meal preserves muscle mass during weight loss. Building meals around lean proteins (chicken, fish, Greek yogurt, legumes), non-starchy vegetables, and moderate amounts of healthy fats (avocado, nuts, olive oil) provides satiety while meeting nutritional needs despite smaller portions.
Exercise enhances peptide effectiveness but isn't strictly required for weight loss, the appetite suppression alone produces significant results. That said, well, combining peptides with structured physical activity optimizes body composition outcomes. Focus on resistance training to preserve muscle mass and moderate cardio for cardiovascular health and additional calorie expenditure.
The synergy between peptides and lifestyle changes works both directions. The medications make dietary adherence easier by reducing cravings and portion sizes naturally. Simultaneously, the healthier habits you establish while taking peptides, meal planning, regular exercise, mindful eating, provide the foundation for maintaining weight loss if you eventually discontinue treatment.

Frequently Asked Questions
How much weight can I realistically expect to lose with peptides like semaglutide?
Clinical trials show FDA-approved peptides like semaglutide produce 12-15% body weight reduction over 68 weeks on average. Individual results vary based on adherence, diet, exercise, and baseline weight, but this represents significantly greater loss than traditional diet pills. Results typically plateau after 1-2 years of treatment.
Are peptides from online sources and compounding pharmacies safe?
Off-label peptides from less-regulated channels carry substantial risks because they lack human clinical data and quality control oversight. The FDA has not verified their purity, potency, or safety profiles. Stick to prescription peptides (Wegovy, Mounjaro, Zepbound) obtained through licensed pharmacies with physician supervision for documented safety.
What's the difference between semaglutide and tirzepatide for weight loss?
Semaglutide (Wegovy) activates only GLP-1 receptors, while tirzepatide (Mounjaro, Zepbound) activates both GLP-1 and GIP receptors for dual-action effects. Tirzepatide appears to enhance fat metabolism and preserve muscle mass more effectively, though both are FDA-approved. Your doctor can help determine which suits your health profile better.
Can I stop taking peptides once I reach my goal weight?
Most patients regain weight after stopping peptide therapy because the underlying appetite regulation and metabolic changes reverse. Some physicians recommend maintenance dosing at lower frequencies, while others transition patients to lifestyle management. Discuss long-term strategy with your doctor before starting treatment.
Are peptides safe for people over 55 with multiple health conditions?
Peptides can be used safely in older adults but require careful medical supervision due to potential drug interactions with common medications and comorbidities like kidney disease or thyroid disorders. Your doctor must review your complete health history and monitor for side effects. Protecting muscle mass through adequate protein and resistance exercise is especially critical for this age group.
What are the most common side effects, and how long do they last?
Gastrointestinal issues (nausea, vomiting, constipation, diarrhea) are most common, typically appearing in the first 2-4 weeks and improving as your body adjusts. Eating smaller meals, staying hydrated, and adjusting fiber intake help manage these effects. Serious side effects are rare with FDA-approved peptides when used under medical supervision.
Do I need to exercise and diet while taking peptides, or do they work alone?
Peptides work best combined with healthy eating and exercise—they're not a standalone solution. They reduce appetite and improve portion control, making diet adherence easier, but you still need to choose nutritious foods and maintain physical activity to maximize fat loss and preserve muscle mass. This combination approach produces superior long-term results.
What's the typical cost, and does insurance cover peptide therapy?
FDA-approved peptides cost $900-$1,500+ monthly without insurance. Most insurance plans cover them only for diabetes management (like Ozempic), not weight loss (Wegovy). Some plans may cover obesity treatment if you meet specific BMI and comorbidity criteria. Check with your insurer and ask your doctor about patient assistance programs to reduce out-of-pocket costs.
